
|
GraPhoBox
|

|
Abstract: The GraPhoBox (Phototropism-Gravitropism
Box) is an experiment, which focuses on phototropism (growth towards a light
source) and gravitropism (growth towards a gravitational vector) in plants.
Both systems have a key role in establishing the basic plant architecture, i.e.
root growth into the soil and shoot growth towards light. Many studies have
been conducted on both systems separately, but never in an integrated approach.
Aim of this experiment is to establish if phototropism and gravitropism in the
model plant Arabidopsis thaliana interact, and to quantify this interaction.
A receptor for phototropism is PHOT1 (also known as NPH1). This mutant exhibits
no phototropism (under specific environmental conditions), but growth and photosynthesis
are unaffected. Gravitropism is initiated by starch sedimentation in plant cells.
Mutants with a deficient starch synthesis, like pgm1, exhibit no gravitropic
response because they have no starch, hence no sedimentation.
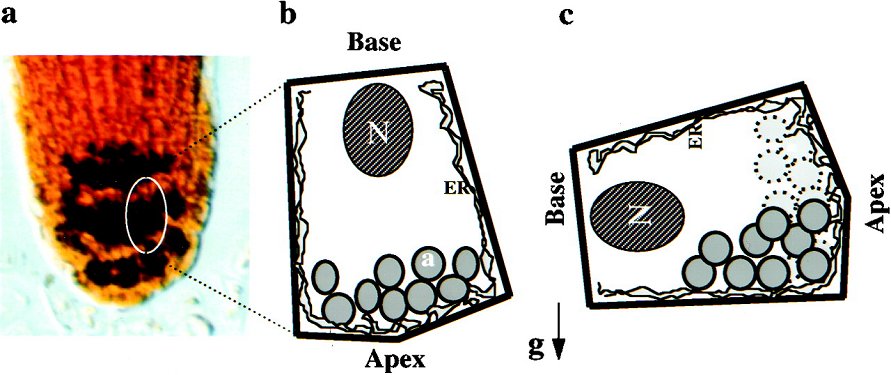 |
Figure 2: Picture (A) and schematic representation of a amylocytes in the root tip of a plant. Note the location of the amyloplasts (a) with respect to the cell and the direction of gravity. N = nucleus, a = amyloplast, ER = Endoplasmatic reticulum, g = gravitational vector. |
The experiment will basically consist of germinating wild type (‘normal’), gravitropic deficient starchless pgm1 mutants, phototropic deficient phot1 mutants and pgm1 phot1 double mutants. These seeds are germinated in different environmental conditions: earth gravity and microgravity, in blue (450 nm) low light conditions and darkness. This will be done in a custom-build, airtight and external light-sealed growth box containing two growth chambers with or without a luminescence system. The experiment takes ten days. By raising the temperature from 5°C to room (cabin/ISS) temperature, germination of the various seeds on agar plates will be initiated. After ten days in space a picture will be taken from the shoots and roots, and the growth angle of both will be measured. Variance analysis will be utilized to see if phototropism and gravitropism have been affected in each set of growth conditions.
Corresponding author:
Karel Buizer (1st year
master student Plant Biology, Utrecht University, Faculty of Biology)
Tel: +31 30 253 3584 / +31 30 253 3172 / +31 30 659 0028
E-mail: k.buizer@students.uu.nl
Introduction: Every higher
plant exhibits a developmental pattern in which the root grows towards the gravitational
vector, and the shoot to a light source. These growth patterns are relatively
strong and visually obvious. The gravitropic root response to gravity allows
the plant to anchor in the soil and take up water and mineral ions and nutrients.
The phototropic response, i.e. the growth towards a light source, allows
the plant to adjust its morphology to gain maximal benefit from the influx of
light to be utilised in the photosynthesis process. These mechanisms are both
observed already by Darwin and his son (1880), but only recently the molecular
background of these observations has been revealed.
Both tropisms have been identified in a number of species, but most studies
discussed the effects in Arabidopsis thaliana. This small weed has an
average life cycle of six weeks, is easily to grow and recently its whole genome
has been sequenced. Because there are lots of mutants available, this plant
is an ideal organism to investigate all kinds of processes, including phototropism
and gravitropism.
Gravitropism has mostly been investigated with the use of clinostats, but barely
in the microgravitational conditions of space. Clinostats are based on a concept
in which the gravitational vector is constantly changed in perspective to the
plant, but after all it is still present. A better alternative is really eliminating
the factor of gravity by conducting experiments in the microgravity of space.
However, due to the limitations of experiments conducted in space (room, crew
time), little is known about possibly different effects between the results
derived from the use of clinostats versus those from “real” microgravity. Although
there are clues that results derived in both conditions follow the same trend,
few experiments, which compare both conditions, have been conducted and little
is known about side effects in using clinostats. However, it has been established
with the use of mutants, clinostats and some space experiments that gravitropism
is the key process in directional development of the root as well as the shoot.
Phototropism is another major component in directional growth of the plant.
It has been established that phototropism occurs mainly in the shoot, but light
has also a negative effect on the direction of root growth. Shoot growth towards
a light source is essential for a plant. Light is essential for photosynthesis
in plants, which supplies the plants with energy-rich compounds. It is critical
to get the most efficient position towards the light source to get the most
gain, and to get an advantage over neighboring plants. This shade avoidance
response is widely spread in the plant kingdom. For roots is a negative phototropism
important, because this means the roots will grow towards the dark (i.e. the
soil) where there are water and nutrients to be found.
Both systems utilize different receptors and signaling pathways, but their effectors
are remarkably the same. Both use differences in cell elongation in the bending
zone as a result of different reactions to auxin. Because gravitropism is one
major component of the directional growth of the plant, it’s obvious to try
to establish if this system influences the effects of phototropism, the other
major directional component. At present it is unclear if both systems are (semi-)
redundant. Although both processes have been readily described in Arabidopsis
thaliana on a phenotypic as well as molecular level, there is almost no
knowledge about the integration of both systems. In this experiment I will investigate
if gravitropism and phototropism are independent systems, and if one system
will compensate loss of function of the other system.
Benefits: The results obtained
from the GraPhoBox experiment can give new insights in the interactions between
the mechanisms underlying the formation of the basic architecture of the plant,
and can also give rise to new idea’s in how plants respond to their environment.
This can have major consequences for future agricultural biotechnology and crop
improvement. Furthermore, the mutual effects of phototropism and gravitropism
resulting from this study can be used as a consideration when new experiments
with plants are designed.
Methods: Two growth chambers with a prism-shape (approximately 12 x 12 x 12 cm) will be constructed and fitted with two agar plates each, mounted perpendicular to each other. One growth chamber will be fitted with a LED (Light Emitting Diode) with a wavelength of 450 nm (blue light) and batteries for power supply. The other growth chamber will be completely dark. All walls are transparent, including the third wall, which allows making a picture of the shoot side of both agar plates. See also Figure 2. During the experiment both growth chambers are sealed from air. External light sources are banned by a Nomex box cover (some kind of foam), which also keeps both growth chambers together. This system, the GraPhoBox, will be kept pre-launch at +5 °C to prevent premature germination. In microgravity the temperature will rise to approximately 20 °C, which is the cabin temperature. This has to be monitored and the temperature of the environment of the earth-based experiments has to be adjusted to be similar to space conditions.
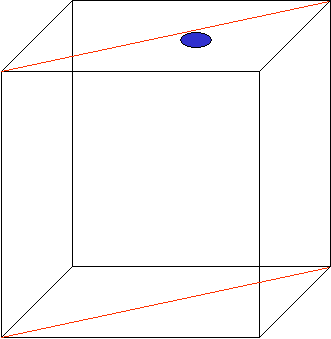 |
Figure 2: Schematic representation of the GraPhoBox. The total box is about 750 gram (max!), approx. 12x12x12 cm, there is one 450 +/- 30 nm LED (blue dot) and 4 agar plates with approx. 40 seeds / mutant. |
After ten days the growth chambers
will be removed from the box, and digital photos are taken from both sides of
each agar plate. From these photos, growth angles will be measured for each
seed strain. When there is less directional growth, the average angle will be
in all cases 0 degrees. But the deviations are much bigger. Thus, by comparing
the deviations of the angles of shoots and roots, the role of both systems can
be established.
In the growth chambers fit two 1 % agar plates containing sufficient nutrients
and water. On this plates are seeds attached from Arabidopsis thaliana.
Each plate has two areas where different mutants and wild-type seeds will germinate.
Each half of the plate has three rows of 15 seeds. Thus a total amount of 45
seeds per mutant and wild type will be germinated per growth chamber. To prevent
the agar plate from disconnecting with its supporting structure, thin bands
will cover the agar plates. This prevents the agar from disconnecting, but allows
the seeds to germinate. This provides also a spatial marker to identify the
various seed strains used on one agar plate
This layout will be adopted for both the experiments conducted in microgravity
as in 1 g conditions.
Acknowledgments: I’d like to thank Dr. Marcel Proveniers and Msc. Bas Rutjens (Molecular Plant Physiology, Utrecht University, Faculty of Biology) for their technical advise in writing the proposal. Rogier Schonenborg for his support from ESA. ESA and the Dutch Ministry of Education, Science and Culture for giving the opportunity to perform the GraPhoBox experiment. The Faculty of Biology in their support for the construction of the growth box.
Related articles:Supplemental information on phototropism and gravitropism can be found at: